



Source: https://pixabay.com/illustrations/dna-analysis-research-3539309/
The Real-Time polymerase chain reaction (RT-PCR) equipment in Centro de Química da Madeira (CQM, University of Madeira) supports research at CQM used as a routine tool for the evaluation of gene expression by CQM researchers from national R&D projects (Post-Doc, PhDs, and Biochemistry (BSc and MSc) and Nanochemistry and Nanomaterials (MSc) students). However, CQM RT-PCR is also open to external services (private and other public institutions).
Submit sample here (exclusively for CQM members) colocar link para submissão de formulário
After the form submission, you will receive an email in your mailbox confirming successful sample registration and the filled form.
If you are not a CQM member and would like to perform a PCR analysis, please contact us via and check the pricing list below.
PCR is a molecular biology technique that allows the amplification of specific DNA sequences, resulting in several copies of the target DNA (or RNA) segments. This principle relies on an initial mixture of the target template DNA, specific primers, nucleotides (dNTPs), ions, and polymerase enzymes that, upon several heating and cooling cycles, promote a three-step cycle of denaturation, annealing, and elongation of the primers.
Denaturation is the first step and involves heating the PCR mixture to a high temperature (approximately 94-98°C), causing the DNA template to denature into two single-stranded chains. The second step, annealing, occurs at lower temperatures (approximately 55-70°C), allowing the short primers to bind to their complementary sequence on each single-stranded DNA template. Finally, in the last step, elongation, the temperature is increased (approximately 68-72°C), promoting the polymerase enzyme to extend the primer strand according to the single-stranded DNA template sequence and synthesise a complementary strand. The end product is a double strand composed of a DNA template chain and a complementary elongated primer. The process is then repeated, usually for up to 40 cycles, resulting in an exponential increase in the number of copies of the target DNA sequence after each cycle.
Advanced molecular approaches have been developed, including real-time PCR, reverse transcription PCR (RT-PCR), and quantitative polymerase chain reaction (qPCR). Real-time PCR takes advantage of fluorescent molecules, making real-time detection of the amplification products possible by increasing the fluorescent signal. Simultaneously, this provides quantitative information (qPCR) on the initial amount of target DNA in the sample. In contrast, RT-PCR allows for the amplification of specific RNA sequences, with a preliminary step where the template DNA is converted into a complementary DNA (cDNA) sequence using reverse transcriptase. This cDNA is then used in the following PCR amplification steps.
PCR is a very fast, sensitive, and specific technique, and its application in modern research includes several scientific fields, such as genetics for genetic mutations and genotyping purposes, diagnostics for viral and bacterial infection detection and treatment, biotechnology to study gene and drug responses for personalised medicine, and forensics, among others.
The equipment was installed in CQM/UMa under its strategic funding project reference: UIDP/00674/2020.

Specifications
| Part number | AIQ060 |
| Product description | 96-well Real-Time PCR instrument with 10.2” touchscreen interface, 6 dye channel filters |
| Sample capacity (wells) | 96 (plates: low-profile, on-skirted, semi-skirted; tubes: low-profile tubes, individual, strips, flat cap; white, frosted or clear and sealed with optical grade film) |
| Reaction volume | 5–150µL (10–50 µL recommended) |
| Excitation source | LED |
| Detection channels | 6 |
| Multiplexing | Up to 6 targets |
| Thermal element | Peltier |
| Max. block ramp rate | 6°C/sec |
| Avg. sample ramp rate | 4°C/sec |
| Temperature uniformity | ±0.2°C |
| Temperature accuracy | ±0.1°C |
| Dye compatibility | SYBR Green, EvaGreen, FAM, VIC, JOE, HEX, CAL Fluor 540, CAL Fluor Orange 560, ROX, TAMRA, TEX615, Quasar 670, CAL Fluor Red 610, Cy5, LIZ, Mustang Purple, Cy5.5, Quasar 705 |
| Custom dye/chemistry | YES |
| Chemistry capability | Fast/Standard |
| Detection sensitivity | 1 copy |
| Sensitivity | Detect differences as small as 1.5-fold in target quantities in single plex reactions |
| Connectivity | USB, Wi-Fi, Ethernet |
Fluorescence channels and dye/probes compatibility
| Channel 1 | Channel 2 |
| Ex 475 ± 14 | Ex 527 ± 10 |
| Em 524 ± 12 | Em 565 ± 12 |
| Dye SYBR™ Green, EvaGreen™, FAM™ | Dye VIC®, HEX™, JOE™, CAL Fluor® 540, CAL Fluor®, Orange 560 |
| Channel 3 | Channel 4 |
| Ex 572 ± 7.5 | Ex 537 ± 13 |
| Em 623 ± 12 | Em 583 ± 11 |
| Dye TAMRA™, Cy3 | Dye ROX™, TEX®615, CAL Fluor® Red 610 |
| Channel 5 | Channel 6 |
| Ex 623 ± 12 | Ex 655 ± 7.5 |
| Em 676 ± 18.5 | Em 711 ± 12.5 |
| Dye CY®5, Quasar®670, Liz®, Mustang Purple® | Dye CY® 5.5, Quasar 705 |
Applications
Gene expression: functional gene products (usually proteins) resulting from a specific gene being transcribed or “expressed”, as a result of a response of the cell to its environment. After DNA transcription, RNA must be transcribed into a functional protein. Because of the involvement of gene expression in disease processes, several molecular methods have been developed to detect very small amounts of DNA and RNA. Using fluorescence technology, such as the most commonly used markers TaqMan and SYBR green, small changes in gene expression can be detected and quantified.
Genotyping involves identifying differences in genetic information between individuals, tracking strains in crop and plant breeding, and tracking the presence of a transgene in transgenic animal models. It is also used in forensics to assess the probability that a specific individual is the source of a DNA sample and in kinship analysis, such as paternity testing. It is often used to detect known variations in specific genes or identify new ones that may be associated with a specific phenotype through copy number variations, insertions, deletions, or single base pairs (SNPs).
Library quantification: Next-generation sequencing (NGS) is a large-scale DNA sequencing technology used to determine DNA and RNA sequences. It aids researchers in studying genetic variations related to diseases, identifying pathogens, and studying tumour subclones. To perform NGS, a sequencing library is created by randomly fragmenting genetic material and adding adapter sequences. The quantification of libraries is essential to ensure equal representation before pooling and optimal dilution for sequencing. qPCR is the recommended method for library quantification owing to its precision, sensitivity, and ability to quantify amplifiable molecules.
Pathogen detection: Detection of the presence of pathogens is important. In recent years, molecular methods, such as real-time PCR (qPCR), have become increasingly popular for pathogen detection because they can offer accurate detection without the need for much time and resources. As many pathogen genomes have been sequenced, qPCR primers specific for groups of pathogens or even a specific pathogen can allow qPCR to serve as a diagnostic tool. Time is often critical for pathogen detection, particularly with regard to patient outcomes. qPCR allows for the detection, quantification, and typing of pathogens much faster than the traditional culture methods.
Multiplex qPCR: refers to having numerous primer sets within a single PCR mixture to evaluate multiple targets in one sample. To differentiate between the products, target-specific primers with different fluorophore labels were used, and each amplification product was detected using a different fluorescent channel. One benefit of multiplexing is that multiple probes can be used in one sample. Multiplexing also ensures minimal crosstalk between the fluorescent dye/probe owing to similar excitation or emission wavelengths. Traditionally, end-point multiplex PCR analyzes the final gene products by gel electrophoresis. This required the final PCR products to be different sizes to be visibly distinguishable from each other on the gel. Using different fluorescent probes eliminates the need for different sizes of PCR products because instead of visually evaluating the size difference, the amount of fluorescence is analysed. Because each PCR product is associated with a specific fluorescent probe, the amount of fluorescence detected in a sample signifies that the target PCR product was present and to what degree.
User fees are intended to cover CQM real-time PCR expenses, which include analysis, consumables, parts, and maintenance.
Choose the class that is best suited to your case or consult us using the address:
The price of the services provided is divided into three classes:
Experimental plan of real-time PCR (gene expression)
1 – RNA extraction from samples using the total RNA isolation kit and/or TRIzol method, quantification, and quality assessment using a NanoDrop spectrophotometre.
2 – Reverse transcription (cDNA synthesis), quantification, and quality assessment.
3 – PCR primers. Each primer set is validated for specificity by qPCR and dissociation curve analysis (SYBR green-based qPCR).
4 – Set up the real-time RT-qPCR reactions using the purified cDNA and run in triplicate qPCR alongside controls (ctrl) and housekeeping (hk) gene, SYBR green-based qPCR.
5 – Data analysis. The average value is normalised to the hk gene selected by the applicant for each sample. To evaluate the results, the ΔΔCt or ΔΔCq method is used to compare changes in gene expression between the controls and samples.
Data analysis is performed using the qRT-PCR data analysis software. The primer sequences, raw Ct values, and analysed data, including all fold-change information, are included in the report.
6 – Use of CQM equipment for PCR runs, reagents not included. Samples previously prepared by the user; ready-to-run.
| Services | Timeline | Pricing (€/sample)* | ||
| Class 1 | Class 2 | Class 3 | ||
| #1 | 2 days | 8.50 | 42.50 | 85 |
| #2 | 1 day | 6.50 | 32.5 | 65 |
| #3 | 2-4 weeks | 30 | 60 | 100 |
| #4 | 3 days | 30 | 60 | 100 |
| #5 | 1 day | 50 | 80 | 120 |
| #6 PCR run
(€/well, reagents not included) |
0.5 | 2.5 | 5 | |
| *Prices for services #1-4 include the cost of material and reagents.
Service #4 prices are per sample per gene of interest, including ctrl and one hk (in triplicate) Service prices are volume-dependent. Prices valid from September 2023; VAT not included, please add tax at the rate of 22% |
||||
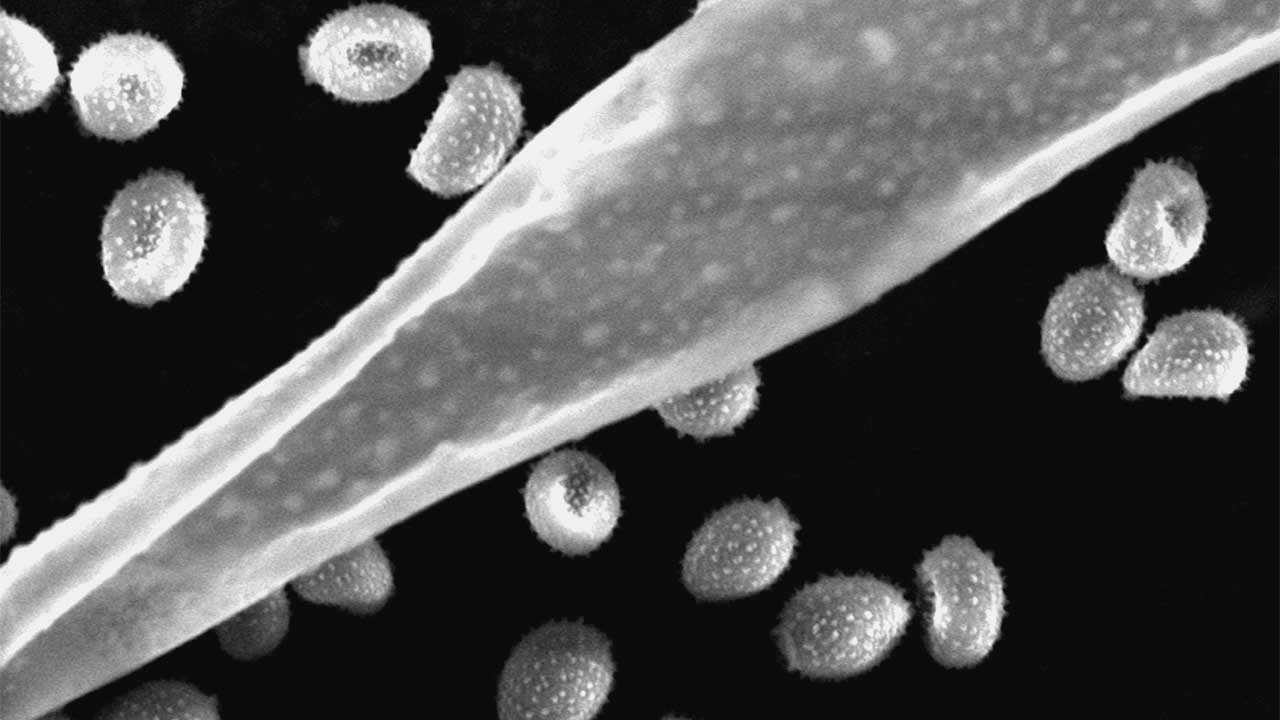

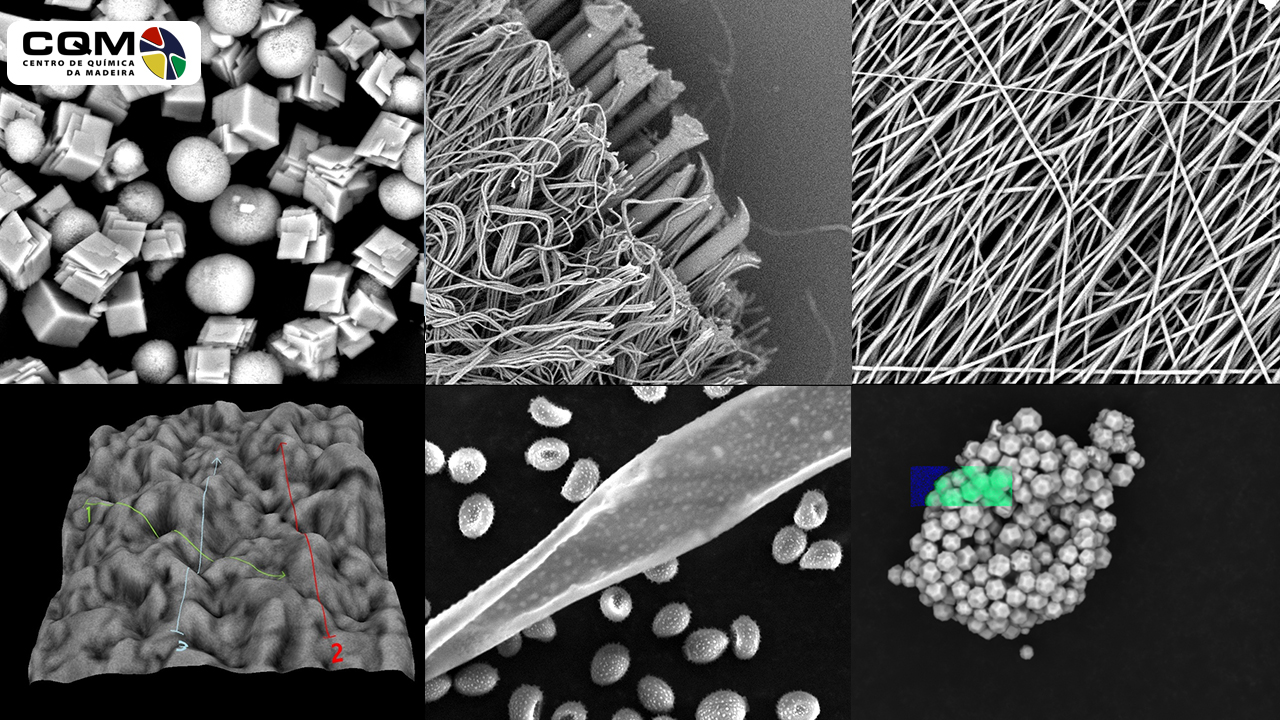
The Scanning Electron Microscopy of the Centro de Química da Madeira (CQM, University of Madeira) main objective is to support research at the Centre, being used as a routine tool for surface characterization of samples surfaces by researchers of CQM from the following national R&D projects; Post-Doc, PhD and Biochemistry (BSc and MSc) and Nanochemistry and Nanomaterials (MSc) students. Furthermore, the CQM SEM is open to external service (private or other public institutions).
Submit sample here (exclusively for CQM members)
In the SEM technique, a focused electron beam causes electron emissions from the sample surface that are measured and spatially imaged. It is commonly used in Materials chemistry, in Chemistry, and Biochemistry for the structural characterization of surfaces; topography and/or quantification of chemical compounds.
The possibility of direct observation of structures through scanning electron microscopy allows an important correlation between processing and properties of materials. The study of the nature of material’s surfaces is relevant for natural products, nanomaterials and many scientific fields such as medicine, environment, microelectronics, or mechanical engineering because many properties of materials are determined precisely by their surfaces and interfaces.
The SEM is routinely used to generate high-resolution images of object’s shapes and to show spatial variations in chemical compositions.
The equipment is installed in CQM/UMa under its strategic funding - Project reference: UID/QUI/00674/2013.
Supervisor: João Rodrigues (PI)
Unit Manager: Rita Castro (PhD)
Autorized User (UT): Rita Castro (PhD)
Applicant: Any researcher, research group or Entity (public or private)
Unit Manager
- Responsible for equipment management, approval of analysis/reports/quotations and consumables aquisition.
- Responsible for equipment operation.

Autorized User
- Responsible for equipment operation.
- Make quarterly reports on the functioning of the Unit. Send to Unit Manager.
- Responsible for the preparation of the equipment reservation/operation time according to the norms of operation of the center.
- Make recommendations to UM about updating or purchasing material in order to ensure an efficient service.
Applicant
- Responsible for time reservation of the equipment and services cost.
- Fill the sample registration form and the registration logbook in accordance with the Center's operating rules.
Bench SEM Microscope Phenom - Pro X
Scanning electron Microscope with qualitative and quantitative analysis of the sample chemical composition by EDS (Energy dispersive X-ray spectroscopy).
EDS specifications
Range of detected elements: from Boron (Z=5) to Americium (Z=95)
Detector:
Lenses
Lighting
Detectors of image
Electronic:
Accessories
Software
Infrastructure Requirements

These Guidelines are intended to be a guide to the autorized users and applicants that uses the CQM SEM equipment and as such must be followed by all. Otherwise, the access to use the microscope will be denied.
The user fees are intended to cover CQM SEM expenses, which includes the analysis, consumables, parts, and maintenance. Minimum time of 1 hour. After 1 hour, the session will be charged in blocks of 30 minutes.
Choose the class that are best suited to your case or consult us using the address: .
The price of services provided is divided into three classes, namely:
|
Class 1 |
Class 2 |
Class 3 |
|
|
Analytical Services (€ / h)* |
€ 35.00 |
€ 61.00 |
€ 101.50 |
|
Specialized sample preparation |
Included in the time of analysis |
||
(prices: valid from January 2022; VAT not included, please add tax at rate of 22%)
* The purchase of service hours packs will benefit of discount. 5% and 10% discount for 10 and 20-hour packs, respectively. Only applied for Classes 2 and 3.
Service Packs
|
Service cost (€ / h) |
Class 1 |
Class 2 |
Class 3 |
|
Pack5 (5 h) |
- |
€ 61.00 |
€ 101.50 |
|
Pack10 (10 h) |
- |
€ 58.00 |
€ 96.50 |
|
Pack20 (20 h) |
- |
€ 55.00 |
€ 91.50 |
(prices: valid from January 2022; VAT not included, please add tax at rate of 22%)
To access the Scanning Electron Microscopy Service, please fill the sample registration form. If you are not a CQM member and would like to perform SEM analysis, please contact us via and check the pricing list.
After the form submission, you will receive in your mail box an email confirming the successful sample registration and the filled form.


The MALDI TOF/TOF MS (Bruker Autoflex maX) equipment installed in CQM, is the first of its kind in the Autonomous Region of Madeira, Portugal. It was acquired with European funds from Programa Madeira 14-20 (PROEQUIPRAM - Reforço do Investimento em Equipamentos e Infraestruturas Científicas na RAM). This equipment is intented for CQM research activities, namely for the molecular mass determination, an also introducing MALDI MS Imaging, searching for the “universal matrix”, involving nanotechnology in MALDI methodology and development of approaches for quantification of molecules. In addition, and depending on the availability of time, the MALDI TOF/TOF MS equipment is open to external services (other public or private institutions).
SUBMIT SAMPLE HERE (for CQM members only)
Equipment: Bruker Autoflex maX Matrix-Assisted Laser Desorption and Ionization (Time-of-Flight) 2 Mass Spectrometer
The Bruker Autoflex maX MALDI TOF/TOF MS is a floor standing equipment with proprietary smartbeam-II™ laser technology with up to 2 kHz repetition rate, high dymanic range FlashDetector™ system in conjuction with a 10 bit 5 Gs/s digitizer and PAN™ wide mass range focusing for unrivaled resolution power and mass accurancy.
Project reference: M1420-01-0145-FEDER-000008
Project Manager: João Rodrigues (PI)
Unit Manager: Rosa Perestrelo (PhD)
Autorised User: Rosa Perestrelo (PhD)
Type of equipament: A – Big equipment
Total investment with taxes: € 403 820.00
CQM acknowledges for the financial support of Programa Madeira 14-20 (PROEQUIPRAM (M1420-01-0145-FEDER-000008) - Reforço do Investimento em Equipamentos e Infraestruturas Científicas na RAM ).

MALDI TOF TOF MS acronym stands for Matrix-Assisted Laser Desorption and Ionization (Time-of-Flight)2 Mass Spectrometry, describing the ionization technique and the type of mass analyzer. The discovery of MALDI ionization in 80´s, independently by two research groups, Tanaka in Japan and joint efforts of Hillenkamp and Karas in Germany, enabled the detection of volatile compounds and high mass biomolecules. It is still a controversy about the who deserved the Nobel prize for MALDI discovery, but the work of both teams was a breakthrough in the field of analyses of biomolecules, particularçy for proteins.
Nowadays, MALDI is still mostly applied for protein analyses, but the number and variety of MALDI applications is expanding (Fig. 1. and Fig. 2.)
Figure 1. Number of applications of MALDI for analyses of individual type of molecules. Source: Web of knowledge database, 19.02.2019.
Figure 2. Number of applications of MALDI for various omics. MALDI is still sought as inevitable source for various omics. Source: Web of knowledge database, 19.02.2019.
MALDI technology uses laser light (UV and/or IR) for ionization and desorption of molecules. “Matrix” is usually an organic aromatic compound, which absorbs the laser energy and transfers it to the analyte, thus minimizing the fragmentation caused by a direct laser hit. Therefore, MALDI is considered a “soft” ionization technique, since the extent of fragmentation of molecules and ions is rather low and some important structural information are preserved. Of course, one can induce fragmentation if more structural information is needed!
There are very few methods, which can be compared to this technology in terms of variety and properties of samples, that can be analyzed. Almost all samples can be analyzed with MALDI, starting with inorganic molecules (low mass), polymers, to high molecular weight proteins and their complexes (in the range of several hundred thousand kDa!), from non polar to highly polar samples, soluble, as well as non soluble, and also samples with high ionization potential.
No derivatization of sample is required. It is simply required to mix the analyte with the matrix or substrate, leave it to co-crystallize or facilitate their crystallization and in a few seconds, you can acquire the spectra!
For most applications, previous separation and purification of samples is also not a pre-requisite for measurements.
The only requirement is that the sample is stable in high vacuum conditions.
Due to its sensitivity, which reaches femtomolar range and small volume of sample required-one can apply 0.2 µL of sample-MALDI is very convenient for samples of biological origin.
MALDI is tolerant against the presence of inorganic salts, and for most applications, there is no need to remove them from the sample.
Please, read these instructions before you prepare and submit the samples for MALDI TOF/TOF mass spectrometry analysis, in order to obtain reliable data. In case of any doubt please contact us at .
MALDI is a very sensitive technique, detecting the analyte up to attomolar range! Since not all substances can ionize equally efficiently, higher amounts are necessary. FOR SOLID/SUSPENSION SAMPLES: If your sample is not soluble in most common solvents, we can still analyze a suspension or in powder. Please submit at least 1 mg, or even more for easier and reliable data, specially for fragmentation pattern analysis. FOR LIQUID SAMPLES: Please submit at least 50 µL of the sample solution at minimum 5 mg/mL and indicate if you need residual amount, storage conditions and the substance's photostability (laser is used for desorption and ionization, therefore, it is better to know for the selection of matrix used).
MALDI is tolerant against the presence of inorganic salts (at physiological conditions) but avoid high salt concentrations and detergents (e.g. Tween 20, SDS or polymers, such as PEG or similar), as these originate high intensity signals, which dominate the spectra and signals from your compound might be suppressed. Please note that your spectrum will reflect the presence of H+, Na+ or K+ adducts, due to the ion formation mechanism. NOTE: If you cannot avoid any of these during the sample preparation, please indicate as detailed as possible the content/composition of the submitted sample.
A screening of sample composition/mixtures is also possible with MALDI, but if you have the presence of easily ionizable species in a mixture, at higher concentrations, it is better to isolate fractions and submit them individually. In the case that you are not able to do that, please indicate or you might wish to consult our operator at LC/MS.
Please, indicate what kind of service you need: do you need simply to confirm the purity of your sample, or you need a complete analysis, description and discussion for potential publication.
The user fees here presented are intended to cover CQM AFM expenses, which includes analysis (and normal use of consumables), technical support, parts and equipment maintenance.
The services are classified by users and type of analysis. Choose the class that are best suited to your case or consult us at the address:
Class 1 - FCT National Centers
Class 2 - Regional and National Public Laboratories
Class 3 - Companies and Laboratories or private Research Centers
| Analysis | Class 1 | Class 2 | Class 3 |
| Acquisition of MS (positive or negative) | € 15.00 | € 25.00 | € 35.00 |
| Additional Analysis (€/sample) | |||
| Acquisition of fragmentation pattern (MS2) | € 15.00 | € 20.00 | € 30.00 |
| Multiple acquisition/testing of conditions | € 5.00 | € 7.00 | € 10.00 |
| Applications of non-standard matrix | € 10.00 | € 15.00 | € 20.00 |
| Description of spectra | €15.00 | € 25.00 | € 30.00 |
| Discussion of results | € 25.00 | € 30.00 | €50.00 |
(Add tax over services with a rate of 22%)
To access the MALDI TOF/TOF MS services, provided by CQM, follow the sample preparation guide and the regulation available above. If you are not a member of CQM and would like to do MALDI TOF/TOF analysis, please contact us at .
SUBMIT SAMPLE HERE (for CQM members only)
After submitting the form, you will receive an email in your mailbox with the indication that your registration was successful and with the filled form attached.
CQM acknowledges for the financial support of Programa Madeira 14-20 (PROEQUIPRAM (M1420-01-0145-FEDER-000008) - Reforço do Investimento em Equipamentos e Infraestruturas Científicas na RAM ).



The NanoDrop One Microvolume UV-Vis Spectrophotometer of Madeira Chemistry Research Center (CQM, University of Madeira) was acquired under the FCT Programmatic Fund UIDP/00674/2020 to improve the facilities and installed capacity of CQM, supporting the research activities included in the different projects underway. This equipment's application areas are mainly in molecular biology, more specifically for the quantification of nucleic acids and proteins. In addition to the support of research activities of CQM, the equipment is open to external service (private or other public institutions) according to the following list of prices:
Price per sample
Price per hour (minimum time: 1h): 20€ (external services only) (plus 22% of taxes).
The fees include sample handling, analysis, and data treatment.
This system provides a quick and easy quantification and assesses purity of samples, such as proteins and nucleic acids. The NanoDrop instrument is able to quantify and qualify DNA, RNA, and protein samples in seconds with only 1-2 µL, and obtain full-spectral data of samples.
Our equipment is the third generation NanoDrop One UV-Vis spectrophotometer and is built with Thermo Scientific™ Acclaro™ Sample Intelligence technology. This technology offers sample contaminant identification, corrected concentrations, and information alerts with guided troubleshooting, helping our researchers to save days of troubleshooting failed experiments.
The following information was adapted from the ThermoFisher Scientific Nanodrop One User Guide (Reference: 269-309102 NanoDrop One UG, Revision B, September 2020).
The Acclaro technology delivers accurate sample quantitative measurements and qualitative information. It is able to identify contaminants in the samples.
To use the NanoDrop service provided by CQM, please contact Dr Mariana Vieira ().
Project Manager: João Rodrigues (PI)
Unit Manager: Mariana Vieira (PhD)
Authorized Users: Mariana Vieira (PhD) and Filipe Olim (MSc)
CQM acknowledges the support of FCT-Fundação para a Ciência e a Tecnologia (Programmatic Fund UIDP/00674/2020).


The Atomic Force Microscopy Nanosurf FlexAFM System of the Centro de Química da Madeira (CQM, University of Madeira), is the first AFM equipment in Autonomous Region of Madeira, Portugal. This equipment is intended for research and has as its main priority the activities/research within the scope of the ECOFIBRAS Project (MAC / 4.6D / 040) for the topographic characterization of fibers of invasive species of Macaronesia, and as part of the research activities of CQM, the study of polymer fibers and DNA films. In addition, and depending on the availability of time, the AFM equipment is open to external services (other public or private institutions).
Submit sample here (exclusively for CQM members)
Atomic force microscopy consists of a type of high resolution microscopy at nanoscale. This technique is used for the analysis of surfaces, in order to obtain information such as topography (roughness, size), material contrast, electrical, magnetic and nanomechanical properties, and surface modification such as lithography. These analyzes can be performed on fixed samples (including biological samples), in air or in liquid medium. The principle of this technique is based on a small probe with a tip (ideally made up of a single atom) coupled to piezoelectric elements with the ability to scan the surface. During this scan, the sensed interactions between the tip and the sample surface atoms are translated into probe deflection / amplitude / frequency variations, which depend on the tip-to-sample distance, that is, whether the interactions are attractive or repulsive. These small variations are then converted, for example, into image, force maps or force curves. The AFM technique has been used in many different areas, such as Biological / Materials Science and Nanomanipulation.
The Nanosurf FlexAFM combined with the C3000 controller is a fully developed research system to perform AFM analysis on its own, or be coupled to inverted microscopes for combination / complementation of results.
AFM System:
- Specification:
X/Y slope correction
Force-distance, amplitude-distance, phase-distance spectroscopy, tip current-tip voltage
Setup Wizard for spectroscopy
- Specifications:
Sample maximum dimensions: 100 mm x 9 mm (width x height)
Manual Approach: 30 mm
Scanning area: 100 μm (XY-range)
Height: 10 μm (Z-range)
Tripod stand-alone scan head with tip scanner
Piezo-based Z-actuator, Optical Z-position sensor and Closed loop Z-control
Low noise photodiode detector (4 quadrant)
Automatic and manual on/off laser (red and near-infrared)
Approach with continuous DC-motor
Automatic self-alignment for probes and magnetic alignment of probe holder
- Operation modes:
Static Force, Dynamic Force, Phase Contrast, Magnetic Force, Electric Force, Basic Spectroscopy, Basic Lithography, Spreading resistance, Force Modulation, Espetroscopia avançada, Litografia avançada
- Specifications:
color (3.1 MP, top view)
monochrome (1.3 MP, side view)
Simultaneous display of top/side view, 4x digital zoom in three steps (1/2/4x)
Informatic system:
Computer HP Pro A Microtower Business
Intel i5 (8th gen) processor
500 GB disc, 4GB RAM.
Monitor/flat screen TFT de 23''
64-bit Windows 10 Professional operating system.
Nanosurf C3000 Controller software v. 3.8
-Provides all functions for the microscope operation during surface analysis, other more advanced operation modes, data analysis for further data processing..
The Scanning Probe Image Processor, SPIP(TM) v. 6.4.4
- Proessing and analysis of microscope images, extraction and analysus of image data.
Gwyddion
- Visualization and analysis of AM data such as dimensions,, image processing, profilometric or topographic data analysis, complex image analysis.
AtomicJ
- Analysis of force data, image and force curves, spectroscopy maps.
Layout Editor
- Graphical vector design for lithography.


CQM's AFM Equipment
Project Manager: João Rodrigues (PI)
Unit Manager: Rita Castro (PhD)
Authorized Users: Rita Castro (PhD)
Unit Manager:
Authorized Users/researchers:
Applicant:
These Guidelines are intended to be a guide to the authorised users and applicants that uses the CQM AFM equipment and as such must be followed by all. Otherwise, the access to use the microscope will be denied.
The user fees here presented are intended to cover CQM AFM expenses, which includes sample preparation, analysis, data treatment, technical support, parts and equipment maintenance. Minimum time request of 5 hours. After 5 hours, the session will be charged in blocks of 1 hour.
The services are classified by users and type of analysis. Choose the class that are best suited to your case or consult us at the address:.
The price of services provided is divided into three classes, namely:
| AFM Analysis type | Class 1 | Class 2 | Class 3 | |
| Basic AFM | Topography and Force Spectroscopy | € 56.00 | € 77.50 | € 86.50 |
| Advanced AFM | Magnetic, Conductive | € 64.00 | € 86.50 | € 94.50 |
| Lithography | € 68.00 | € 90.50 | € 98.50 | |
| Special Probe | Operation in Liquid | € 68.00 | € 90.50 | € 98.50 |
| High Resolution (air / liquid) | € 100.00 | € 117.00 | € 127.00 | |
(prices: valid from January 2022; VAT not included, please add tax at rate of 22%).
* Minimum time: 5h. The purchase of service hours packs will benefit of discount. 5% and 10% discount for 20 and
30-hour packs, respectively. Only applied for Classes 2 and 3.
To access the Atomic Force Microscopy service, provided by CQM, follow the sample preparation guide and the regulation available above. If you are not a member of CQM and would like to do AFM reviews, please contact us at .
Submit sample here (exclusively for CQM members)
After submitting the form, you will receive an email in your mailbox with the indication that your registration was successful and with the filled form attached.
The Principal Investigator and CQM acknowledges for the financial support of the MAC 2014-2020 Territorial Cooperation Program through the European Regional Development Fund (FEDER) under the ECOFIBRAS Project (Sustainable exploitation of Macaronesia invasive plant species for industrial fibers, MAC/4.6 D/040).